
WCD2 by pro aqua
Reliable Process Water Cleaning for Heater-Cooler Units
Less cleaning.
More perfusion


Managing Process Water Quality in Heater-Cooler Units
Microbiological contamination of heater-cooler units (HCU) process water by pathogens such as Pseudomonas aeruginosa, Legionella pneumophila, and Mycobacterium chimaera requires reliable hygiene control and typically needs recurring chemical disinfection procedures.
WCD2 supports reliable HCU process water hygiene through short automated treatment cycles integrated into routine technical workflows, reducing the need for recurring manual chemical disinfection. This helps lower personnel workload, improve device availability, and minimize chemical exposure and consumables dependency in clinical environments.
The Problem
Pathogens in HCU Process Water
Microbiological contamination of HCU process water can represent a potential health risk factor and requires reliable quality control. Pseudomonas aeruginosa, Legionella pneumophila, and Mycobacterium chimaera have been associated with contaminated process water and related infections.
Demand for Extensive Cleaning
Maintaining hygienic process water conditions requires recurring cleaning and disinfection procedures involving strong chemical agents such as hydrogen peroxide, peracetic acid, and chlorine-based disinfectants, which may damage the HCU and even pose occupational health risks for staff.
High Operational Resource Demand
Conventional cleaning protocols require substantial perfusion staff time and can take several hours per device each month. They also reduce HCU availability, increase documentation effort and operational costs, and may contribute to long-term component wear due to repeated chemical exposure.
Substantial Economic Impact
Downtime, personnel workload, occupational exposure risks from repeated handling of chemical disinfectants, recurring consumables, and lifecycle effects on system components create operational and financial pressure for clinical institutions and can affect long-term service planning for HCU fleets.
The Solution:
WCD2 - Automated Process Water Treatment for HCUs


WCD2 is an electrochemical process water quality system for HCUs. It treats process water using boron-doped diamond electrode technology and operates in short automated routine cycles as part of regular technical workflows. The system supports stable microbiological process water conditions and reduces the need for recurring manual chemical disinfection routines.

Key Features
WCD2 Advantages at a Glance

Automated process water cleaning & disinfection
Fully automated treatment process is performed with a single button press within 10 minutes

Patented electrochemical oxidation technology
Oxidative agents are generated directly from process water using patented boron-doped diamond electrodes

Compatible with common HCUs
The WCD2-cHCU1 and WCD2-cHCU2 are designed for using with widely used HCU types

Maintains pathogen-free process water conditions
The WCD2 treatment keeps HCU process water free from microbial contamination
Integration into existing HCU infrastructure
WCD2 connects externally to compatible HCU and treats the entire process water volume without technical adjustments

Automated process documentation support
All treatment processes are automatically recorded as part of reproducible management routines
How It Works
System preparation: WCD2 operation requires a microbiologically clean HCU following baseline decontamination. A small amount of WCD2 salt mixture is added to the process water to establish the conductivity required for electrochemical treatment.
Step 1
Connecting the Hoses
For process water treatment, the existing HCU hoses are disconnected from the attached components and connected directly to the WCD2 without modification of the system. No additional tools or equipment required.
Step 2
Starting the Treatment
The complete process water is routed through the WCD2 flow cells. Water in additional connections, such as drain lines, can also be treated, ensuring that water often missed in conventional procedures is also cleaned.
Step 3
10 Minutes long Cleaning
Reactive oxidants are generated electrochemically in the WCD2 flow cells which inactivate and break down microorganisms, germs and pathogens across the entire process water which ensures its quality.
Result
Disinfected Process Water
The entire process water system remains permanently pathogen-free, ensuring stable and reliable operating conditions.

The Technology
Meet our patented Boron-Doped Diamond (BDD) Electrode Technology

Industrial-grade flow cells with more than 20 years of experience
WCD2 uses BDD electrode flow cells that have been successfully used in industrial water treatment applications for more than 20 years
Versatile electrochemical oxidation capability
BDD flow cells allow the treatment of a wide range of contaminants, from microorganisms to persistent substances such as PFAS and other organic residues
In situ generation of highly reactive oxidants directly from water
As water passes through the flow cell, highly reactive species are generated at the electrode surface and immediately inactivate germs in the process water
Highly durable electrode material for continuous technical operation
The diamond electrode surface is chemically inert and resistant to corrosion and ageing, ensuring adequate oxidation performance over long operating periods
The Evidence
Selected Findings from Published and Institutional Studies
University Hospital Regensburg Study
• 60 microbiological sampling series over an 11-month investigation period • No detection of Pseudomonas aeruginosa during the study period • Legionella remained below the technical detection limit throughout the investigation • One Mycobacterium chimaera finding was associated with the sampling outlet and was not detected again after technical modification Source: Foltan M, Nikisch A, Dembianny J, et al. A solution for global hygienic challenges regarding the application of heater-cooler systems in cardiac surgery. Perfusion. 2021. DOI: 10.1177/02676591211033483.
Laboratory Validation
Institute for Hygiene, Medical University of Graz • Under the stated test conditions, all selected test organisms were reduced to below the detection limit after 2.5 minutes • Selected test organisms included Pseudomonas aeruginosa, a staphylococcal species, and Bacillus subtilis • Testing was performed under defined laboratory conditions in duplicate Source: Mascher F. Test Report: Inactivation of selected test bacteria in water by treatment with a pro aqua diamond electrode. Institute for Hygiene, Medical University of Graz. 09.02.2017.
Material Compatibility
SEM Surface Analysis and Long-Term Material Assessment • Oxygenators from two manufacturers were exposed to treated water for 36 hours • No perforations or micro-ruptures were observed on the examined surfaces • After 11 months of constant application, no degradation marks were found on the cooling heat exchanger or heating coil Source: Foltan M, Nikisch A, Dembianny J, et al. A solution for global hygienic challenges regarding the application of heater-cooler systems in cardiac surgery. Perfusion. 2021. DOI: 10.1177/02676591211033483.
Austrian Federal Office for Safety in Health Care (BASG)
• Administrative decision issued on 27 November 2023 • BASG determined that WCD2 is not a medical device within the meaning of Regulation (EU) 2017/745 • BASG also determined that WCD2 is not an accessory to a medical device under Regulation (EU) 2017/745 • The decision is based on the stated intended purpose of WCD2 as a system for cleaning and disinfecting process water in hypothermia devices, not the medical device itself Source: Austrian Federal Office for Safety in Health Care (BASG). Administrative Decision - Demarcation “WCD2 - Water Cleaning & Disinfection Device”. Reference number GZ: 102523930 / 3569224-008. 27.11.2023.
Application Refusal under MDR - TÜV SÜD Product Service GmbH
Formal refusal of the MDR application for initial certification of WCD2-c, dated 8 October 2024 • The refusal concerns an application for conformity assessment under Regulation (EU) 2017/745 • TÜV SÜD explicitly states that it did not overrule the BASG decision of 27 November 2023 • The refusal is based on the BASG classification that WCD2 is not a medical device or medical device accessory under MDR Source: TÜV SÜD Product Service GmbH. Refusal of Application Due to Regulation. Project number 713342194-AR. 08.10.2024.
Regulatory Assessment - Graz University of Technology
• The stated intended purpose of WCD2 is the automatic disinfection of water in the water circuits of hypothermia devices • The report clearly distinguishes between disinfection of process water and disinfection of the medical device itself • Based on the stated intended purpose, the report concludes that WCD2 is not a medical device within the meaning of MDR 2017/745 Source: Graz University of Technology. Test Report 01/23 - Classification of WCD2 under MDR 2017/745. 22.02.2023.
Regulatory Classification
Your Benefits
Less Cleaning. More Perfusion.

Automated treatment
-
Starts with a single button press
-
Runs unattended
-
Consistent treatment every day
Reduced maintenance time
-
10 minutes of automated cleaning per day
-
Runs while staff performs other tasks
-
No interruption of perfusion routines
Increased HCU availability
-
Predictable 10-minute daily routine
-
Minimal device downtime
-
Immediate readiness
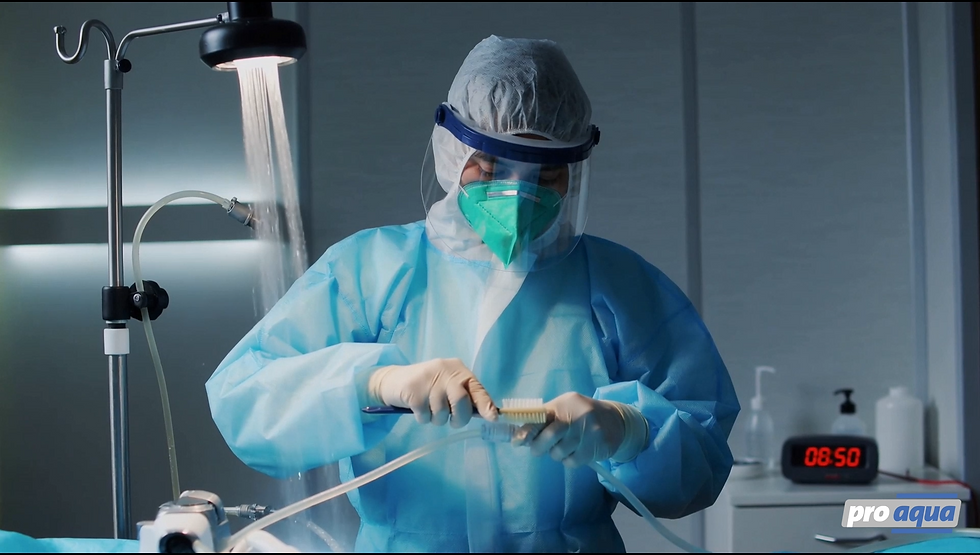
Safe working conditions
-
In situ process water disinfection
-
Low active agent concentrations
-
Occupational safety

Lower consumables dependency
-
Reduced need for disinfectants
-
Reduced procurement effort
-
No dedicated infrastructure required

Structured documentation
-
Automated recording of treatment cycles
-
Reproducible routines
-
Transparent process history

Improved material compatibility
-
Operation with low oxidant concentrations
-
Reduced long-term component stress
-
No recurring shock disinfection exposure

Same operation for multiple HCUs
-
Operator-independent treatment
-
Predictable routines across installations
-
Scalable integration into service structures
